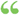 If, when a drug is sold under, or by a name recognized in the United States Pharmacopoeia, or National Formulary, it differs from the standard of strength, quality, or purity, as determined by the... If, when a drug is sold under, or by a name recognized in the United States Pharmacopoeia, or National Formulary, it differs from the standard of strength, quality, or purity, as determined by the... 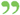 The Modern family physician v. 1 - Page 264by Kenelm Winslow - 1915Full view The Modern family physician v. 1 - Page 264by Kenelm Winslow - 1915Full view - About this book
 | 1909 - 520 pages
...internal or external use;" and >e?tion 7 of that act provides that a drug s'ull be deemed adulterated "if when a drug is sold under or by a name recognized...standard of strength, quality, or purity as determined ly the test laid down in the Unilecl States Pharmacopceia or National Formulary official at the time... | |
 | Max C. Fleischmann College of Agriculture. Agricultural Experiment Station - 1913 - 576 pages
...7. Drugs shall be deemed adulterated within the meaning of this Act in any of the following cases: First — If, when a drug is sold under or by a name...Formulary, it differs from the standard of strength or purity as determined by the tests laid down in the United States Pharmacopoeia or National Formulary... | |
 | California State Board of Health - 1914 - 1204 pages
...the Food and Drugs Act states that "an article shall be deemed to be adulterated: In ease of drugs: If, when a drug is sold under or by a name recognized...Formulary, it differs from the standard of strength, qrality. or purity, ns determined by the test laid down in tho United States Pharmacopeia or National... | |
 | Kentucky. State Board of Health - 1919 - 316 pages
...ti"nl"tu" of drug shall be deemed to be adulterated: First. If, when a drug is sold under or by the name recognized in the United States Pharmacopoeia...strength, quality or purity, as determined by the tests laid down in the United States Pharmacopoeia or National Formulary official at the time of investigation.... | |
 | New York (N.Y.). Department of Health - 1922 - 924 pages
...internal use, or both. Drugs as herein denned shall be deemed adulterated: (1) If, when sold by or under a name recognized in the United States Pharmacopoeia...from the standard of strength, quality, or purity, as stated in the United States Pharmacopoeia or National Formulary at the time of investigation. (2) If... | |
 | New York (N.Y.). Department of Health - 1915 - 364 pages
...internal use, or both. Drugs as herein denned shall be deemed adulterated: (1) If, when sold by or under a name recognized in the United States Pharmacopoeia...from the standard of strength, quality, or purity as stated in the United States Pharmacopoeia or National Formulary at the time of investigation. (2) If... | |
 | 1922 - 978 pages
...provides that an article shall be deemed to be adulterated, in the case of drugs, if when sold under a name recognized in the United States Pharmacopoeia...differs from the standard of strength, quality or purity therein laid down, or if its strength or purity fall below the professed standard or quality... | |
 | 1909 - 764 pages
...adulterated : First. If, when a drug is sold under or by a name recognized in the United States Pharmacopeia or National Formulary, it differs from the standard...strength, quality, or purity as determined by the tests laid down in the United States Pharmacopoeia or National Formulary official at the time of investigation:... | |
 | 1907 - 472 pages
...shall be deemed to be adulterated : In case of drugs : First. If, when a drug is sold or dispensed under or by a name recognized in the United States Pharmacopoeia or National Formulary, it differs in composition, or standard of strength, quality or purity, recognized by the United States Pharmacopoeia,... | |
 | 1920 - 986 pages
...United States, the fourth edition of the National Formulary, or the American Homeopathic Pharmacopoeia, it differs from the standard of strength, quality, or purity as determined by the test or formula laid down in the ninth revision of the Pharmacopoeia of the United States, the fourth edition... | |
| |